ORG is independently owned and physician-led. We add locations where we find strong investigators and patient populations underrepresented in clinical research.
Your investigators focus on patients. We handle regulatory, IRB, eSource, data entry, and coordination – every visit, every study.
Dr. Scott Phillips, Indiana ENT, with no prior trial experience, has enrolled over 70 patients in a Pfizer pediatric migraine study – a result of experienced operational support and access to the right patient populations.
Eight sites across Indiana and Arizona, reaching patient populations with limited access to clinical trials.
Options Research Group advances medical knowledge through the rigorous conduct of clinical trials. We support sponsors, investigators, and study participants with efficient start-up, disciplined enrollment, and reliable data collection – operating to the highest standards of ethical conduct, regulatory compliance, and patient safety.
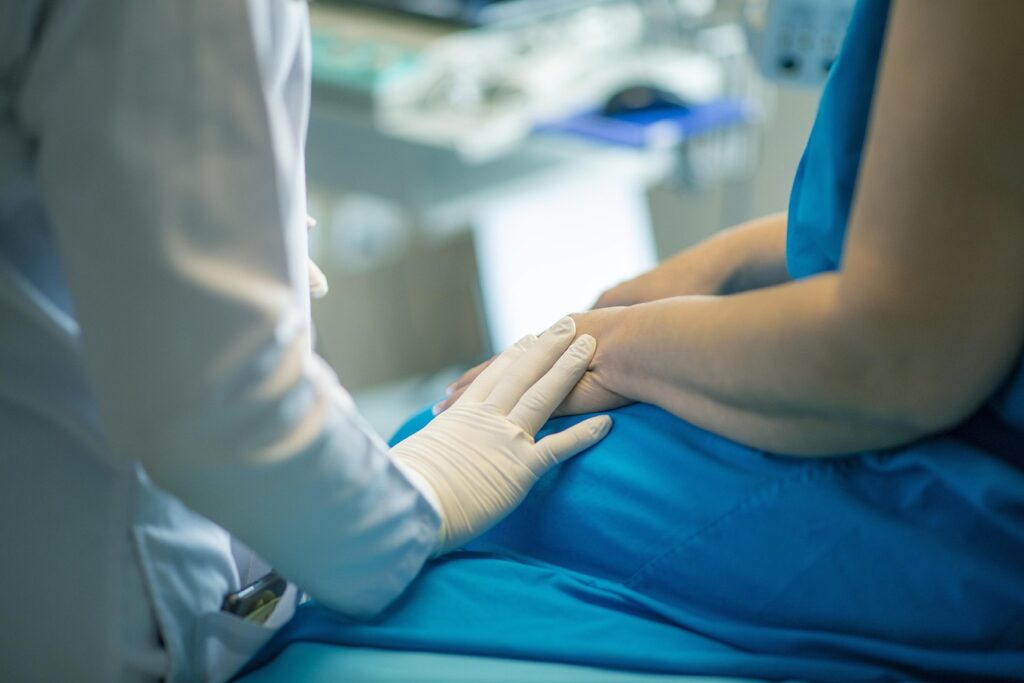
We manage the start-up process – IRB submissions, informed consent preparation, regulatory document collection, and site activation – so investigators are trial-ready without administrative burden.

Our dedicated research staff handles ongoing study management including protocol compliance monitoring, serious adverse event reporting, timely IRB submissions, recruitment advertising, and site readiness for sponsor and CRA visits.

From patient scheduling and follow-up to contract negotiations, document archiving, and trial close-out – we handle the operational work so investigators can focus on patient care.
 Co-Founder
Co-FounderMaureen Banning RN, CCRC
Co-Founder, Director of Clinical Operations

Ryan Banning
Co-Founder, CEO

Viviana Gaytan
Clinical Research Coordinator

Chayton Krueger
Clinical Research Coordinator

Marcie Sears
Clinical Research Coordinator

Kevin Gibbons
Clinical Research Coordinator

Kathleen Paz, BS
Regulatory Specialist

Elena Paz
Data Management

Jason Babcock, MBA, ACRP-CP
VP of Business Operations


Options Research Group is exceptionally well connected within the industry and well positioned to pair investigators and support staff with the right study at the right time. Their support for our many needs – including documentation, procedures, and potential pitfalls – is exemplary, providing comprehensive assistance with integrity and enthusiasm. As a principal investigator for multiple studies, I feel that the support and attention to detail given to me, the study sponsors, and, above all, the study participants are impeccable.

Options Research Group bridges the gap between providers and pharmaceutical companies by bringing their expertise to the table. Their knowledge base starts with study start-up and seamlessly follows through to the completion of a trial. I have peace of mind knowing that trials performed at my site are conducted ethically and according to protocol. I know that my patients are in great hands.

Prior to working with Options Research Group, I had no experience in the field of pharmaceutical research. From day one, they guided and trained me to become a sub-investigator. It brings me joy to know that my patients are so well cared for, and it adds excitement to my medical practice to have additional treatment options to offer my patients.

I am continually impressed with Options Research Group’s commitment to excellence in clinical research. Since becoming a sub-investigator ten years ago, I have consistently seen the team go above and beyond in the time, care, and attention they devote to our patients. Their professionalism, organization, and patient-first approach make a meaningful difference in both the quality of the research conducted and the overall experience for those who participate in our studies.
We select locations where strong investigators serve patient communities with limited access to clinical trials. Each site operates with the same team, same standards, and same operational support.
Westfield
ENT, Pediatrics, Hematology, Oncology, Internal Medicine
Carmel
ENT, Flight Medicine, Pediatrics, Oncology
Indianapolis
Orthopedics, Primary Care, Critical Care, Pulmonary
West Lafayette
Dermatology
Also in: Noblesville, Zionsville, Lafayette
Phoenix
Pediatrics, Neurology, Dermatology
Coming 2026
Serving Central Phoenix and the greater metropolitan area